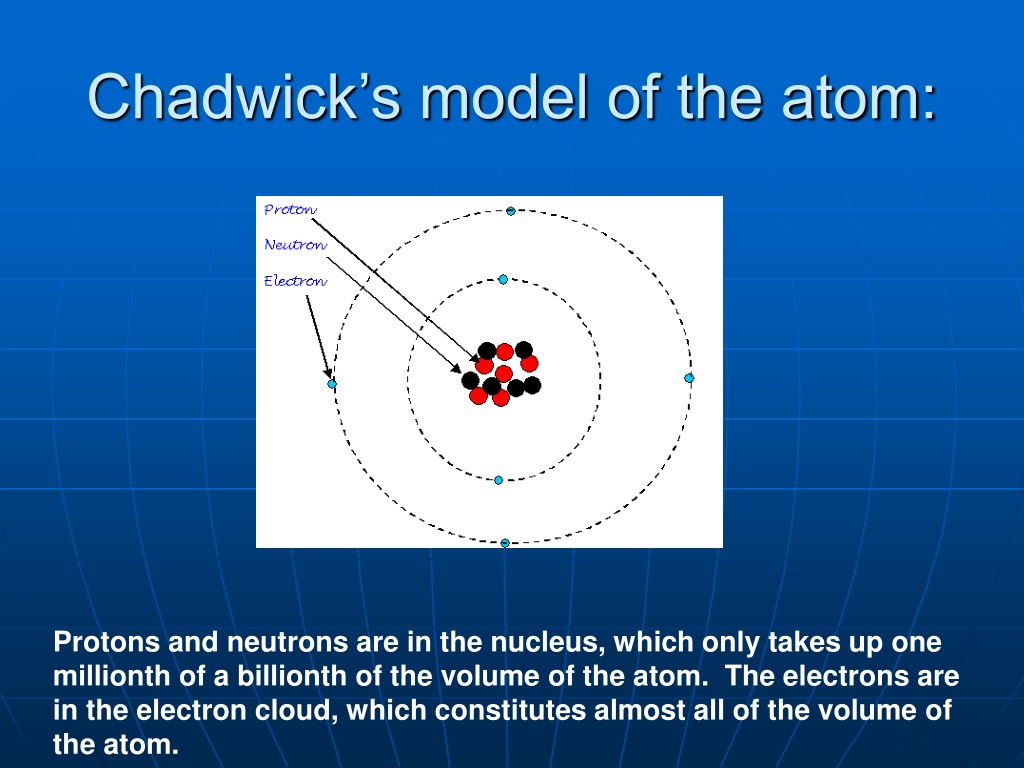 
In 1932, he tried similar experiments himself, and became convinced that the radiation ejected by the beryllium was in fact a neutral particle about the mass of a proton. Photons, having no mass, wouldn’t knock loose particles as heavy as protons from the target, he reasoned. Joliot-Curie believed the radiation hitting the paraffin target must be high energy gamma photons, but Chadwick thought that explanation didn’t fit. They found that this radiation knocked loose protons from hydrogen atoms in that target, and those protons recoiled with very high velocity. One experiment in particular caught his attention: Frédéric and Irène Joliot-Curie had studied the then-unidentified radiation from beryllium as it hit a paraffin wax target. Chadwick had noticed some odd features of this radiation, and began to think it might instead consist of neutral particles such as those Rutherford had proposed. Some scientists thought this highly penetrating radiation emitted by the beryllium consisted of high energy photons. Around 1930, several researchers, including German physicist Walter Bothe and his student Becker had begun bombarding beryllium with alpha particles from a polonium source and studying the radiation emitted by the beryllium as a result. In 1920, Rutherford proposed that an electron and a proton could actually combine to form a new, neutral particle, but there was no real evidence for this, and the proposed neutral particle would be difficult to detect.Ĭhadwick went on to work on other projects, but kept thinking about the problem. Some scientists thought there were additional protons in the nucleus, along with an equal number of electrons to cancel out the additional charge. For instance, helium was known to have an atomic number of 2 but a mass number of 4. However, it seemed there must be something in the nucleus in addition to protons. Rutherford had discovered the atomic nucleus in 1911, and had observed the proton in 1919. In 1923, Chadwick was appointed assistant director of Cavendish Laboratory. Chadwick was able to continue to work on radioactivity, now with more sophisticated apparatus than tin foil and toothpaste. Using some tin foil and wood he built an electroscope and did some simple experiments.Īfter the war, Chadwick returned to England, where he finished his PhD in Cambridge in 1921 with Rutherford, who was then Director of Cambridge University’s Cavendish laboratory. Though many chemicals were hard to get hold of, Chadwick even found a type of radioactive toothpaste that was on the market in Germany at the time, and managed to persuade the guards to supply him with it. To keep from being bored, he and some fellow prisoners formed a science club, lectured to each other, and managed to convince the guards to let them set up a small lab. This did not entirely stop his scientific studies. Unfortunately, not long after he arrived, WWI broke out and Chadwick ended up spending the next four years in a prison camp there.  
In 1914, Chadwick decided to travel to Germany to study with Hans Geiger. He was a shy child from a working class family, but his talents caught his teachers’ attention, and he was sent to study physics at the University of Manchester, where he worked with Ernest Rutherford on various radioactivity studies. Chadwick was born in1891 in Manchester, England.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
